The First Aid Beauty Face Moisturizer Recall has sent shockwaves through the skincare community, affecting thousands of loyal customers who rely on the brand’s cult-favorite Ultra Repair Cream. This comprehensive analysis examines the details, impact, and broader implications of the First Aid Beauty Face Moisturizer Recall that began in December 2024.
Understanding the First Aid Beauty Face Moisturizer Recall
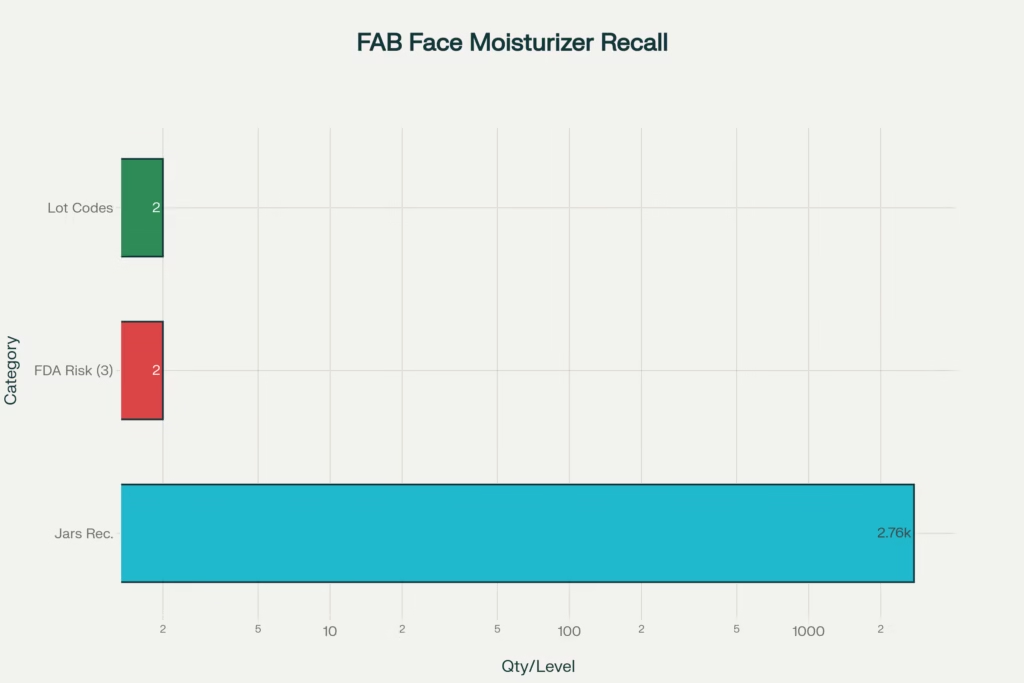
The First Aid Beauty Face Moisturizer Recall centers on 2,756 jars of Ultra Repair Cream in the Coconut Vanilla scent that were mistakenly released to consumers. On November 20, 2024, First Aid Beauty discovered that products “intended for quarantine” had been inadvertently sold through their website. This discovery triggered what would become a significant First Aid Beauty Face Moisturizer Recall affecting customers nationwide.
The FDA classified this First Aid Beauty Face Moisturizer Recall as Class II on January 21, 2025—the second-highest risk level in the agency’s classification system. This classification indicates that the First Aid Beauty Face Moisturizer Recall involves products that “may cause temporary or medically reversible adverse health consequences,” though the probability of serious harm remains remote.
First Aid Beauty Face Moisturizer Recall Key Statistics (2025)
Timeline of the First Aid Beauty Face Moisturizer Recall
The First Aid Beauty Face Moisturizer Recall unfolded over several months:
- November 20, 2024: First Aid Beauty identifies the issue with quarantined products being sold
- December 23, 2024: Official First Aid Beauty Face Moisturizer Recall initiated voluntarily
- January 21, 2025: FDA formally classifies the First Aid Beauty Face Moisturizer Recall as Class II
- January 22-25, 2025: Major media outlets report extensively on the First Aid Beauty Face Moisturizer Recall
Product Details in the First Aid Beauty Face Moisturizer Recall
The First Aid Beauty Face Moisturizer Recall specifically affects:
| Recall Detail | Specification |
|---|---|
| Product | Ultra Repair Cream, Colloidal Oatmeal 0.5% |
| Scent | Coconut Vanilla only |
| Size | 14-ounce jars |
| Affected Lots | 24D44 (exp 4/10/2026), 24D45 (exp 4/11/2026) |
| Total Units | 2,756 jars |
| Distribution | FirstAidBeauty.com primarily |
| Retail Value | Approximately $104,728 (at $38 per jar) |
The First Aid Beauty Face Moisturizer Recall does not affect other scents or sizes of the Ultra Repair Cream product line.
Why the First Aid Beauty Face Moisturizer Recall Happened

The First Aid Beauty Face Moisturizer Recall resulted from a deviation in Current Good Manufacturing Practice (CGMP) regulations. According to FDA documents, the issue occurred because “product intended for quarantine was inadvertently distributed”.
CGMP regulations are critical safety protocols that ensure products meet quality and safety standards before reaching consumers. The First Aid Beauty Face Moisturizer Recall was triggered not by confirmed contamination, but by a process failure that violated these essential quality control measures.
Consumer Impact of the First Aid Beauty Face Moisturizer Recall
The First Aid Beauty Face Moisturizer Recall has had significant emotional and practical impacts on consumers:
Emotional Response to the First Aid Beauty Face Moisturizer Recall
Many customers expressed anxiety and concern about the First Aid Beauty Face Moisturizer Recall on social media platforms. Ultra Repair Cream has a dedicated following among people with eczema and sensitive skin, making the First Aid Beauty Face Moisturizer Recall particularly distressing for those who depend on the product.
“For years, this cream was my go-to for soothing irritated skin. This recall made me double-check every jar I had…” — Reddit user discussing the First Aid Beauty Face Moisturizer Recall
Brand Response to the First Aid Beauty Face Moisturizer Recall
First Aid Beauty responded proactively to the First Aid Beauty Face Moisturizer Recall by:
- Directly contacting all affected customers via email
- Providing replacement products in different scents (often Grapefruit)
- Offering clear instructions to stop use and discard affected products
- Maintaining transparency throughout the First Aid Beauty Face Moisturizer Recall process
Also Read : Vegamour Review Beauty Lifestyle Skin Makeup Blog: The Science-Backed 2025 Guide
Statistical Analysis of the First Aid Beauty Face Moisturizer Recall
The First Aid Beauty Face Moisturizer Recall represents a relatively small portion of the brand’s overall sales volume. First Aid Beauty claims to sell one jar of Ultra Repair Cream every ten seconds, which would translate to approximately 9,000 jars monthly across all variants. The First Aid Beauty Face Moisturizer Recall of 2,756 jars therefore represents roughly 30% of one month’s estimated sales for the entire Ultra Repair Cream line.
First Aid Beauty Face Moisturizer Recall by the Numbers
| Statistic | Value | Impact Level |
|---|---|---|
| Jars in First Aid Beauty Face Moisturizer Recall | 2,756 | Moderate |
| FDA Risk Classification | Class II (2 of 3) | Significant |
| Customer Response Rate | Nearly 100% contacted directly | High |
| Geographic Impact | Nationwide (US only) | Broad |
| Financial Impact | ~$104,728 retail value | Substantial |
| Time to Resolution | 2 months from discovery | Standard |
Medical and Safety Implications of the First Aid Beauty Face Moisturizer Recall
The Class II designation of the First Aid Beauty Face Moisturizer Recall indicates specific health risk parameters. According to FDA guidelines, this classification means:
- Temporary or medically reversible adverse health effects are possible
- The probability of serious adverse health consequences is remote
- The product deviation represents a regulatory compliance issue requiring correction
Dr. Chad Hartman, a dermatologist affiliated with US Dermatology Partners, emphasized in previous interviews that moisturizers serve as protective barriers for skin health. However, he also noted that some ingredients—particularly fragrances like those in the First Aid Beauty Face Moisturizer Recall Coconut Vanilla variant—can cause irritation in sensitive individuals.
Industry Context of the First Aid Beauty Face Moisturizer Recall
The First Aid Beauty Face Moisturizer Recall occurs within a broader context of increased FDA oversight of the cosmetics industry. Since 2023, new legislation has expanded regulations and tightened reporting obligations for manufacturers, leading to more frequent product recalls.
Research from MasterControl indicates that FDA and Consumer Product Safety Commission recalls increased significantly between 2018 and 2022, with the First Aid Beauty Face Moisturizer Recall representing part of this trend toward enhanced consumer protection.
What Consumers Should Do About the First Aid Beauty Face Moisturizer Recall
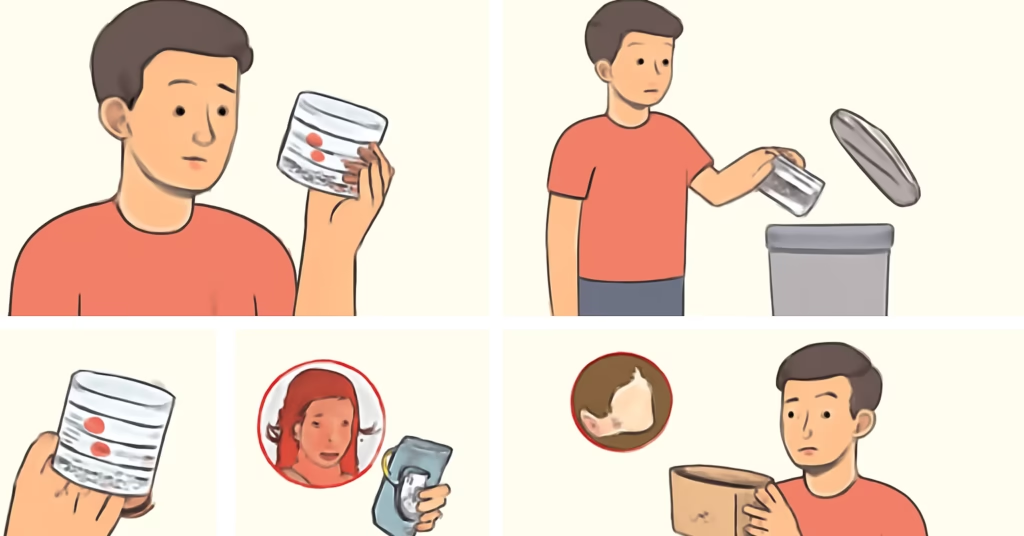
If you own Ultra Repair Cream, here’s your action plan for the First Aid Beauty Face Moisturizer Recall:
Step 1: Check Your Product
Examine the bottom or back of your jar for:
- Coconut Vanilla scent designation
- 14-ounce size
- Lot codes 24D44 or 24D45
- Expiration dates April 10, 2026 or April 11, 2026
Step 2: If Your Product Matches the First Aid Beauty Face Moisturizer Recall
- Stop using the product immediately
- Discard the jar safely
- Contact First Aid Beauty customer service at customerservice@firstaidbeauty.com or 800-322-3619
- Request a replacement or refund
Step 3: Monitor for Any Reactions
If you’ve used a product affected by the First Aid Beauty Face Moisturizer Recall and experience skin irritation, discontinue use and consult a healthcare professional, especially if you have sensitive skin or eczema.
Also Read : Skin Better Science: Your Complete Guide to Revolutionary Medical-Grade Skincare
Alternative Products Following the First Aid Beauty Face Moisturizer Recall
The First Aid Beauty Face Moisturizer Recall has prompted many consumers to seek alternatives. Dermatologists recommend considering:
- Fragrance-free options: To avoid potential irritation that led to concerns in the First Aid Beauty Face Moisturizer Recall
- Colloidal oatmeal formulations: Similar to the active ingredient in the recalled product
- Eczema-friendly moisturizers: For those who relied on the recalled product for sensitive skin conditions
Popular alternatives mentioned by skincare experts include CeraVe Moisturizing Cream, La Roche-Posay Toleriane Double Repair Face Moisturizer, and Vanicream Daily Facial Moisturizer.
Long-term Implications of the First Aid Beauty Face Moisturizer Recall
The First Aid Beauty Face Moisturizer Recall may have several lasting effects:
For the Brand
- Increased quality control measures
- Enhanced quarantine and release procedures
- Potential impact on consumer trust and loyalty
- Possible regulatory scrutiny of other products
For the Industry
- Reinforcement of CGMP compliance importance
- Increased awareness of process control requirements
- Potential for more frequent voluntary recalls as preventive measures
For Consumers
- Greater awareness of lot codes and expiration dates
- Increased vigilance regarding product recalls
- Enhanced understanding of FDA classification systems
Expert Opinions on the First Aid Beauty Face Moisturizer Recall
Skincare experts and dermatologists have generally praised First Aid Beauty’s handling of the First Aid Beauty Face Moisturizer Recall. The proactive approach, direct customer communication, and voluntary nature of the recall demonstrate industry best practices for managing product safety issues.
However, some experts note that the First Aid Beauty Face Moisturizer Recall highlights the importance of robust quality control systems in cosmetics manufacturing, particularly for products marketed to sensitive skin populations.
Lessons Learned from the First Aid Beauty Face Moisturizer Recall
The First Aid Beauty Face Moisturizer Recall offers several important lessons:
For Manufacturers
- Quality control processes must be followed rigorously
- Voluntary recalls can demonstrate commitment to consumer safety
- Direct communication with affected customers is essential
- Transparency throughout the recall process helps maintain trust
For Consumers
- Always check lot codes when recalls are announced
- Register products with manufacturers when possible for recall notifications
- Understand FDA classification systems to assess risk levels
- Keep receipts and purchase records for skincare products
For Regulators
- Clear classification systems help consumers understand risk levels
- Process deviations require serious attention even without confirmed contamination
- Industry cooperation in voluntary recalls benefits public safety
Moving Forward After the First Aid Beauty Face Moisturizer Recall
The First Aid Beauty Face Moisturizer Recall represents a significant event in the skincare industry, but it also demonstrates how modern regulatory systems and responsible manufacturer practices can protect consumers. The brand’s quick response, the FDA’s clear classification, and extensive media coverage have ensured that affected customers received timely and accurate information.
For skincare enthusiasts, the First Aid Beauty Face Moisturizer Recall serves as a reminder of the importance of staying informed about product safety issues and understanding how to respond when recalls occur. The incident doesn’t necessarily reflect poorly on the overall quality of First Aid Beauty products, but rather highlights the complex manufacturing processes that ensure product safety and the vigilance required to maintain quality standards.
Frequently Asked Questions
- Does this affect the fragrance-free Ultra Repair Cream?
Current reporting and brand statements indicate the recall is limited to the Coconut Vanilla 14oz jars in lots 24D44 and 24D45. - I bought mine at Sephora/Ulta/Target—am I affected?
Coverage differs by outlet; some reports say the variant was sold exclusively on FirstAidBeauty.com, while others mention broader distribution language tied to the brand and product line overall—use your lot code to decide your next step and contact the brand for confirmation. - What if I used the affected product and had no reaction?
The Class II designation implies that serious harm is unlikely; still, discontinue use of affected lots and follow recall guidance. - Will there be more recalls?
There’s no evidence of broader product issues at this time; the classification and notices specifically reference the listed lots and scent. Continue to watch official FDA recall postings and brand communications for updates.
Conclusion: Understanding the First Aid Beauty Face Moisturizer Recall
The First Aid Beauty Face Moisturizer Recall of 2,756 jars of Ultra Repair Cream in Coconut Vanilla represents a significant but well-managed product safety event. With its Class II FDA classification, the First Aid Beauty Face Moisturizer Recall indicates moderate risk levels that require consumer attention but don’t suggest imminent danger.
The comprehensive response to the First Aid Beauty Face Moisturizer Recall—including direct customer notification, product replacement programs, and regulatory cooperation—demonstrates industry best practices for managing product safety issues. For consumers, the First Aid Beauty Face Moisturizer Recall reinforces the importance of staying informed about product recalls and understanding how to respond appropriately.
As the skincare industry continues to evolve under enhanced regulatory oversight, events like the First Aid Beauty Face Moisturizer Recall will likely become more common as manufacturers err on the side of caution to protect consumer safety. The key for consumers is to stay informed, understand risk classifications, and respond promptly to recall notices when they occur.
The First Aid Beauty Face Moisturizer Recall ultimately serves as a case study in effective recall management, demonstrating that transparent communication and proactive consumer protection measures can help maintain trust even when manufacturing issues arise.